List view / Grid view
Therapeutics
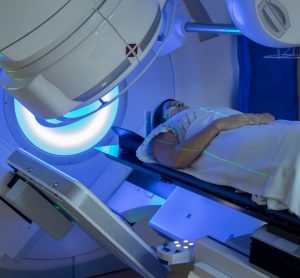
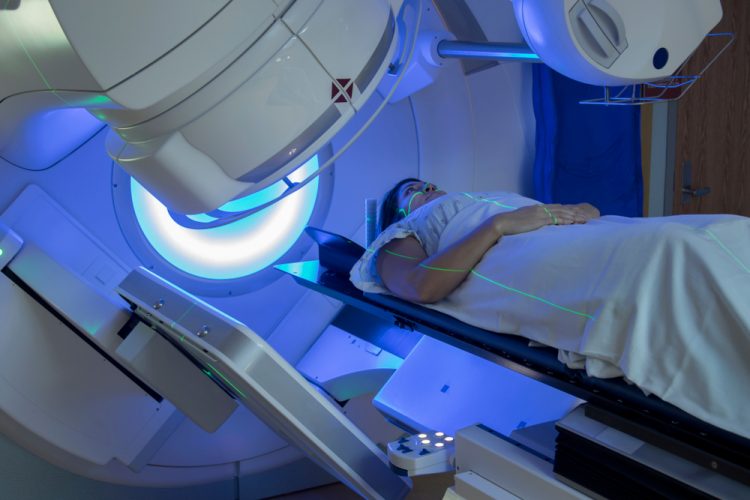
Forxiga approved in EU for chronic kidney disease treatment
Forxiga’s approval, based on unprecedented Phase III data, is the most significant advancement in chronic kidney disease (CKD) treatment in over 20 years.
Libtayo® with chemotherapy trial stopped early due to success
Phase III trial of Libtayo® (cemiplimab) combined with chemotherapy stops early due to significant improvement in overall survival in lung cancer patients.
Collaboration and integration to progress clinical research
Here, Lucinda Cash-Gibson and Francesco Patalano discuss why the pharmaceutical industry is collaborating to develop protocols for patient-centric integrated platform trials and what the clinical research industry may look like in future.
Global API CDMO market to value almost $140bn by 2028
Market research suggests the Asia Pacific region could soon overtake Europe and North America in the active pharmaceutical ingredient (API) contract development and manufacturing organisation (CDMO) market.
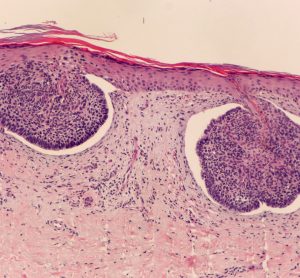
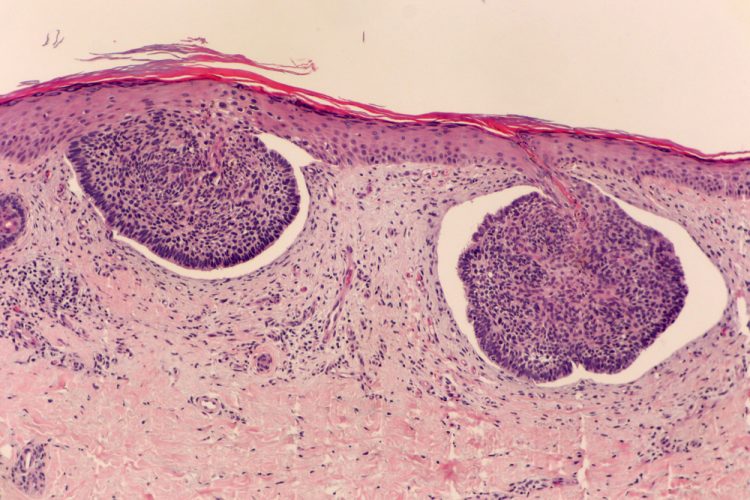
Remetinostat shows promise as a BCC treatment in Phase II study
Remetinostat, a first-in-class histone deacetylase inhibitor gel, displayed signs of clinical efficacy in patients with basal cell carcinoma.
Ritlecitinib improves scalp hair regrowth in alopecia trial
Ritlecitinib was shown to reduce scalp hair loss to less than or equal to 20 percent after six months in Pfizer’s Phase IIb/III trial.
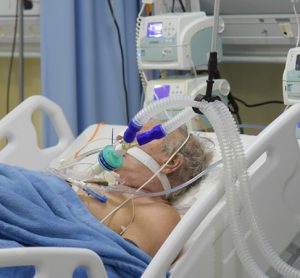
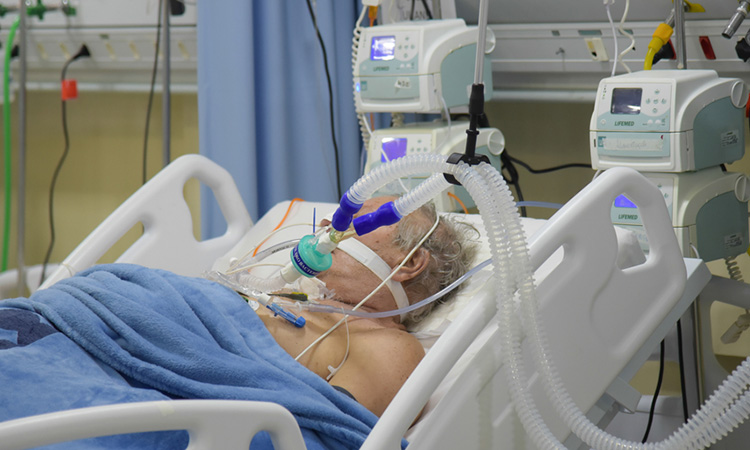
Blood thinners reduce need for organ support in COVID-19 patients
Clinical trial shows that treating moderately ill COVID-19 patients with a full-dose blood thinner reduced their need for organ support.
Bayer to acquire Vividion Therapeutics for novel drug discovery platform
Bayer to pay $1.5 billion upfront to acquire Viridion Therapeutics, thus strengthening its small molecule capabilities and expanding its reach into new modalities.
Global pharma contract manufacturing market to value $120bn by 2027
Growth in the pharmaceutical contract manufacturing market to be driven by advanced technologies, the rising aging population and the increase in chronic disease cases, says report.
BioNTech strengthens neoantigen TCR programme with Kite acquisition
BioNTech acquires Kite’s solid tumour neoantigen T cell receptor research platform and Gaithersburg manufacturing facility for undisclosed amount.
Biogen announces results from Phase IIIb NOVA study for MS
Results from the NOVA study show every six-week dosing with natalizumab is as effective as every four-week in relapsing-remitting MS.
NaviFUS begins clinical trial for brain tumour treatment
NaviFUS® was approved by the Taiwan FDA for use in a new clinical trial for the treatment of terminal primary brain tumours.
Baricitinib reduces mortality of hospitalised COVID patients by 46 percent
New data shows Lilly’s Olumiant (baricitinib) significantly reduces the risk of death in hospitalised COVID-19 patients receiving invasive mechanical ventilation or ECMO.
AstraZeneca reveals $15.54 billion in revenues in H1 2021
H1 financial results show AstraZeneca’s revenues grew by 23 percent in 2021, with $14.37 billion attributed to activities aside from COVID-19 vaccine sales.