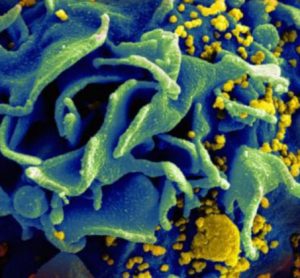
Regular infusions of a novel antibody may suppress HIV for four months
According to a new report, regular infusions of an antibody that blocks the HIV binding site on human immune cells may have suppressed levels of HIV for up to four months in people undergoing a short-term pause in their antiretroviral therapy (ART) regimens.