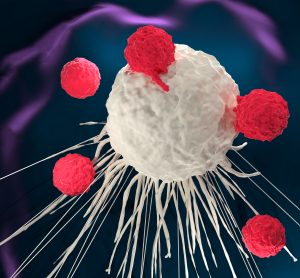
CRISPR therapies could be one of the most transformative in human history, says GlobalData
Adam Pearson, Senior Oncology Analyst at GlobalData, comments on the development of CRISPR-based therapies and outlines potential opportunities and challenges in the space.