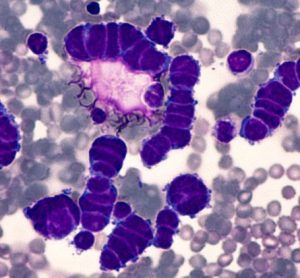
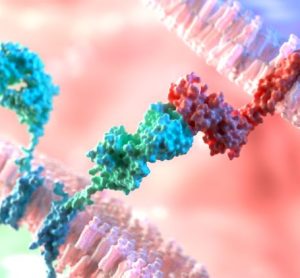
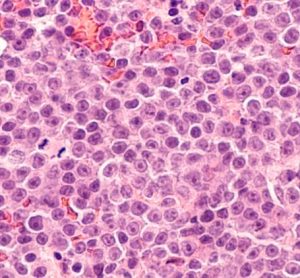
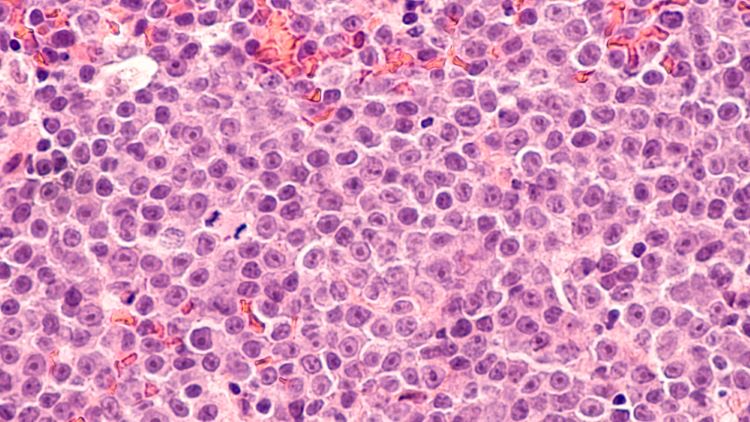
Developing point-of-care CAR T manufacturing
Driven by technological advances there is now increased scope for point-of-care manufacturing of CAR T-cell therapy. Arnon Nagler, Professor of Medicine at Tel Aviv University, discusses key benefits, challenges and lessons learned from developing an in-house programme.