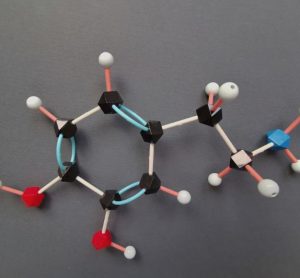
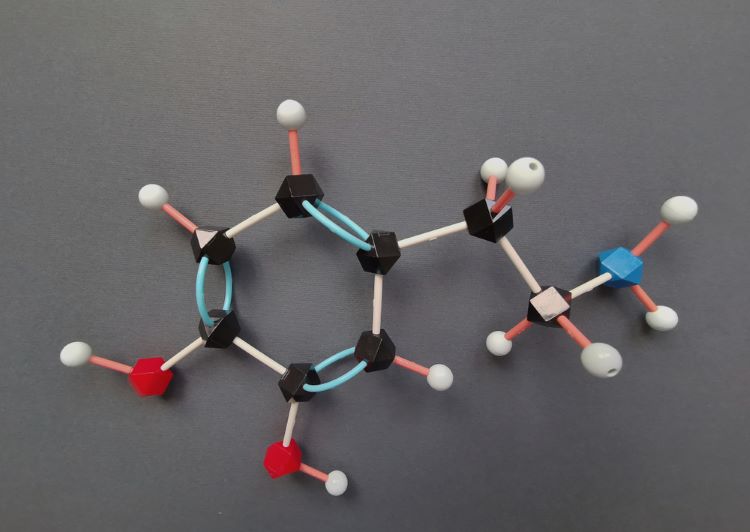
Pharma Horizons: Sustainability
European Pharmaceutical Review’s latest Pharma Horizons report provides insight on key sustainability developments in the pharmaceutical industry and covers topics including regulation, manufacturing and clinical development.