Tracking and tracing to the root cause: case studies in microbial contamination
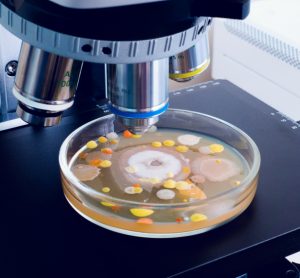
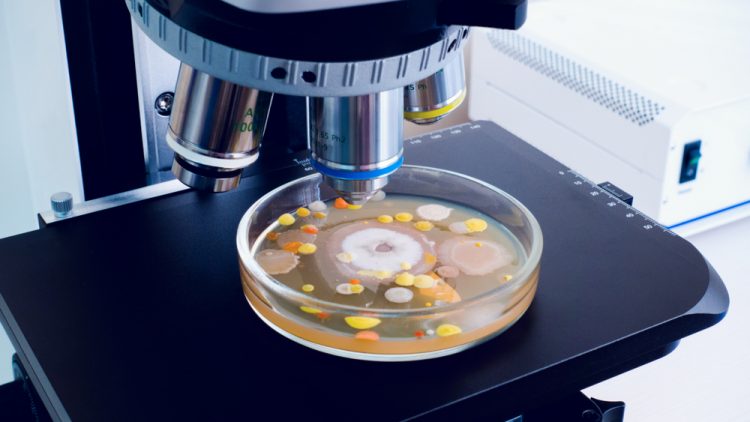
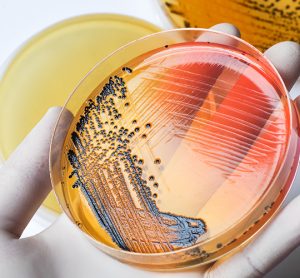
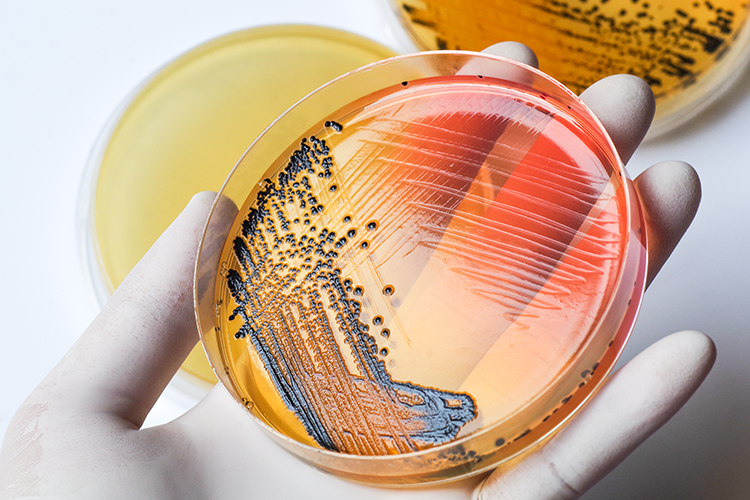
In this article, pharmaceutical microbiologist and contamination control expert Tim Sandle presents three microbial contamination investigation case studies, highlighting the key lessons for pharmaceutical microbiologists to take away and the underlying importance of identifying the root cause of microbial data deviations.