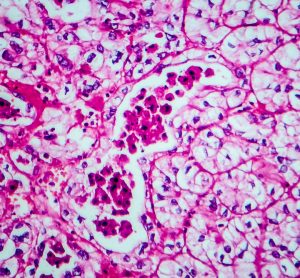
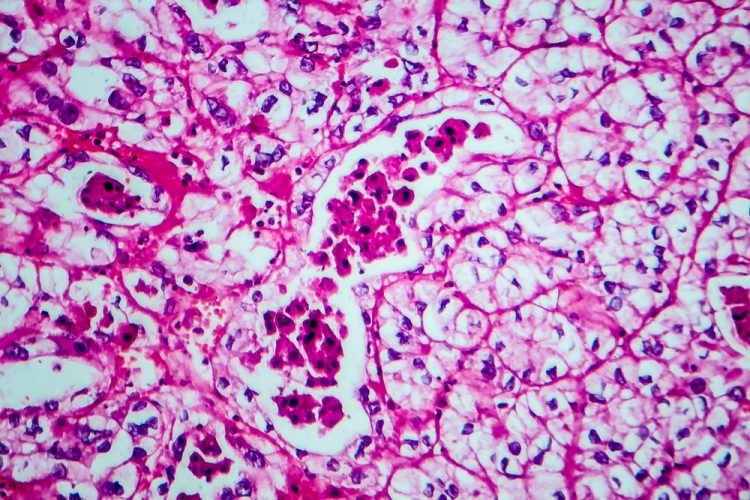
Prototype AI test could predict promising cancer drug combinations
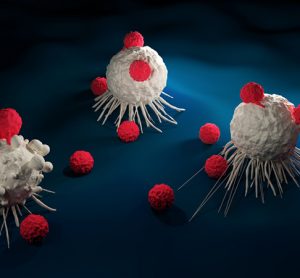
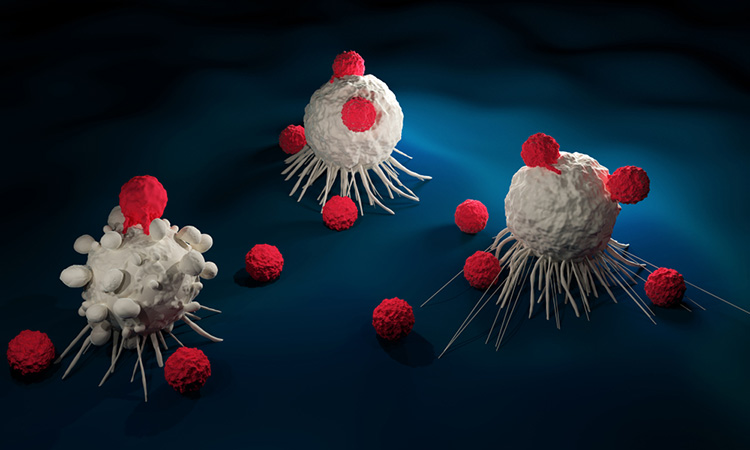
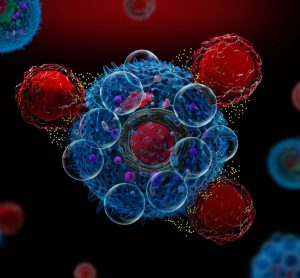
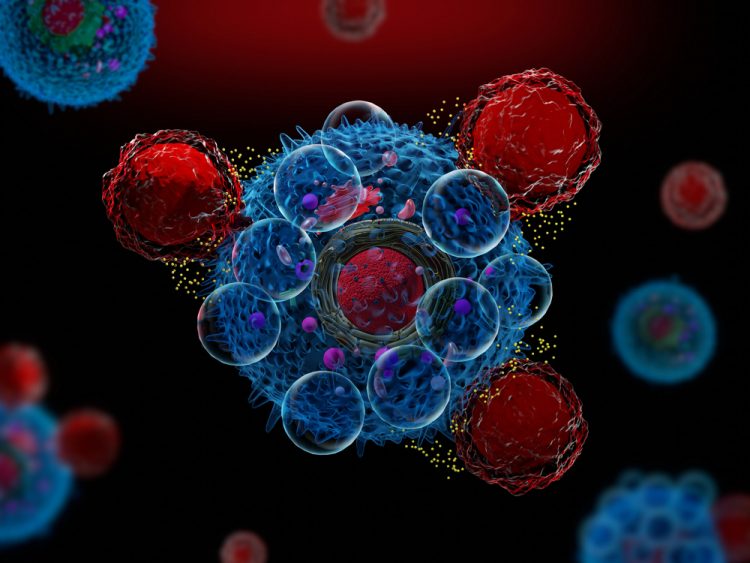
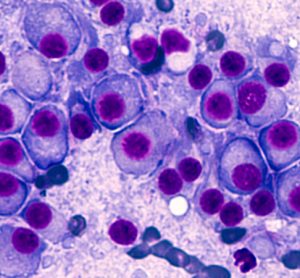
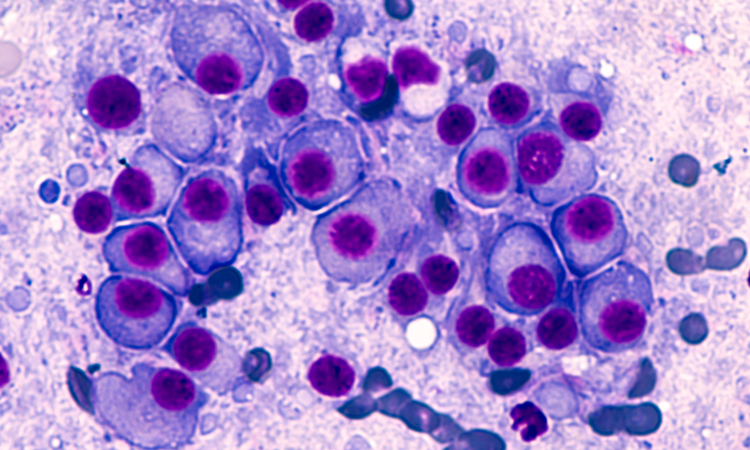
An artificial intelligence (AI)-based test was able to predict cancer patients’ response to drugs more accurately than is currently possible with genetic testing, finds proof-of-concept study.



![Merck logo atop Merck Research Laboratories campus in Silicon Valley [Credit: Michael Vi / Shutterstock.com].](https://www.europeanpharmaceuticalreview.com/wp-content/uploads/Merck-5-300x278.jpg)
![Merck logo atop Merck Research Laboratories campus in Silicon Valley [Credit: Michael Vi / Shutterstock.com].](https://www.europeanpharmaceuticalreview.com/wp-content/uploads/Merck-5-e1648202271156.jpg)


![Takeda logo on a building in San Diego, California 2020 [Credit: Michael Vi/Shutterstock.com].](https://www.europeanpharmaceuticalreview.com/wp-content/uploads/Takeda-1-300x278.jpg)
![Takeda logo on a building in San Diego, California 2020 [Credit: Michael Vi/Shutterstock.com].](https://www.europeanpharmaceuticalreview.com/wp-content/uploads/Takeda-1-e1641995614300.jpg)