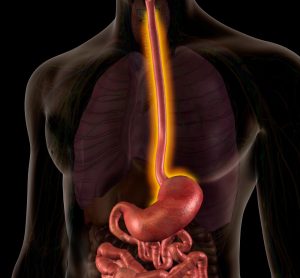
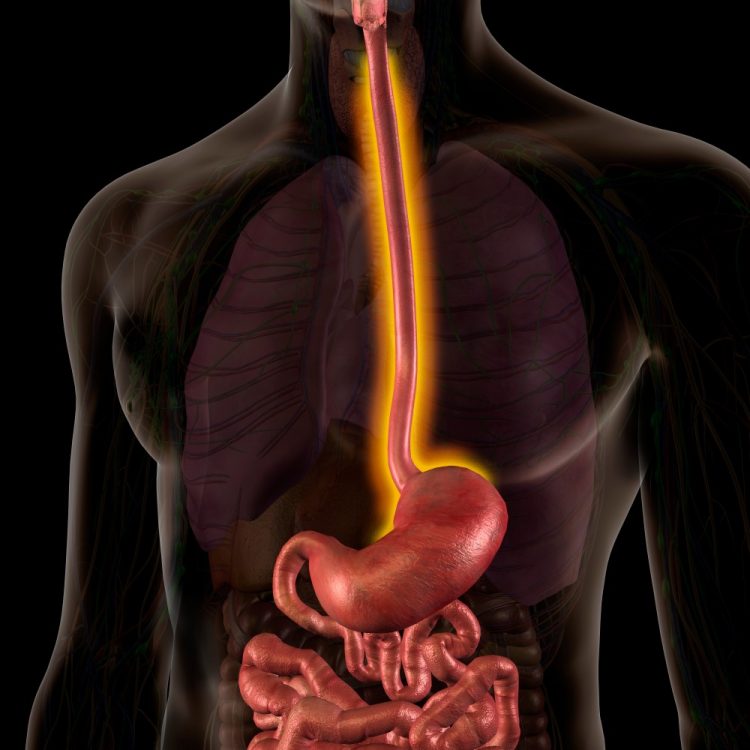
Dupixent® delivers histological remission for paediatric EoE patients
Phase III trial data shows Dupixent® (dupilumab) has the potential to improve signs of eosinophilic oesophagitis (EoE) and support healthy weight gain in children with the condition.
















![WuXi Biologics logo on a lit up phone screen [Credit: Piotr Swat/Shutterstock.com].](https://www.europeanpharmaceuticalreview.com/wp-content/uploads/WuXi-Biologics-300x278.jpg)
![WuXi Biologics logo on a lit up phone screen [Credit: Piotr Swat/Shutterstock.com].](https://www.europeanpharmaceuticalreview.com/wp-content/uploads/WuXi-Biologics.jpg)

