FDA approves Boehringer Ingelheim’s Praxbind
19 October 2015 | By Victoria White
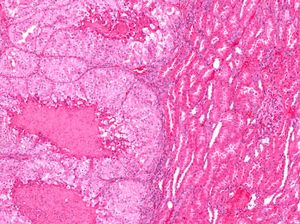
Praxbind is indicated for patients treated with Pradaxa, when reversal of the anticoagulant effects of Pradaxa is needed for emergency surgery or in uncontrolled bleeding...