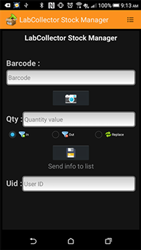
Smaller and faster: Mira M-3 handheld Raman spectrometer for material verification
6 February 2017 | By Metrohm
The Mira M-3 is the new handheld Raman spectrometer from Metrohm. Barely larger than a smartphone, the Mira M-3 enables true single-handed operation...