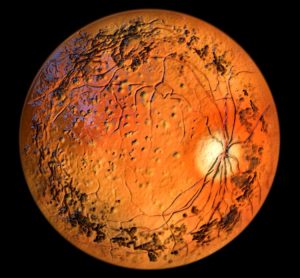
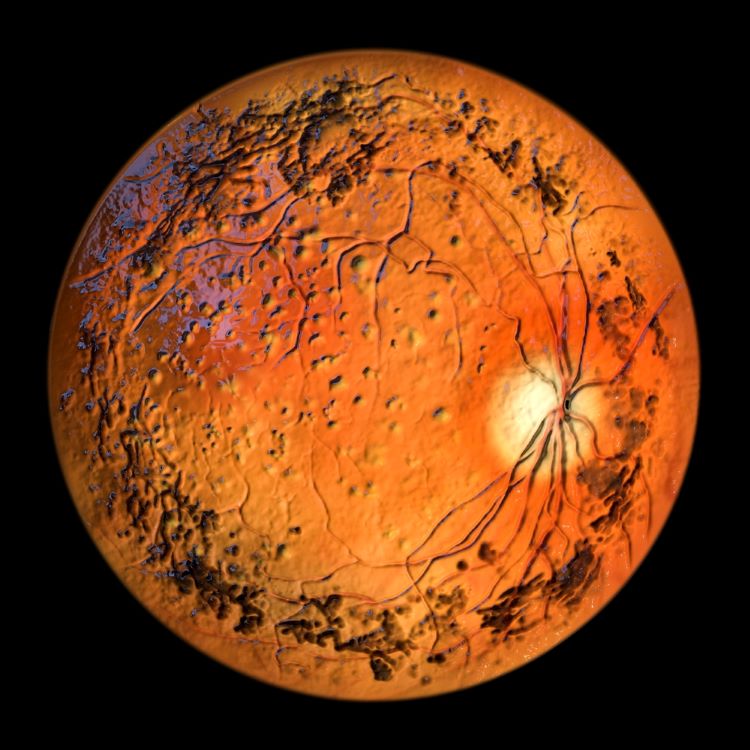
First-in-human data for gene therapy signals potential in childhood blindness
Developed and manufactured under a Manufacturer’s ‘Specials’ Licence (MSL), the gene therapy could provide a novel treatment option for paediatric patients at the earliest stage of the rare eye disease.