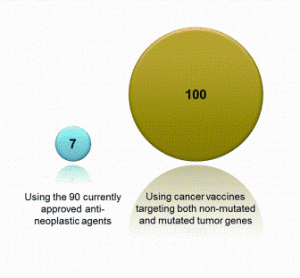
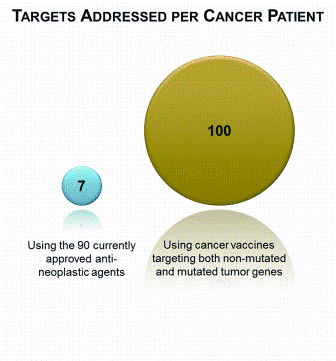
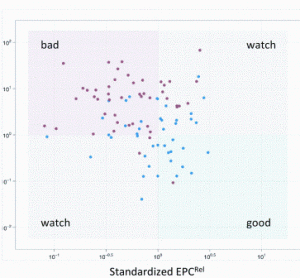
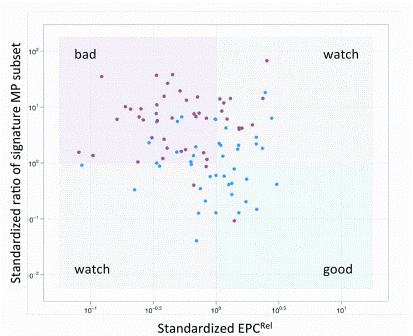
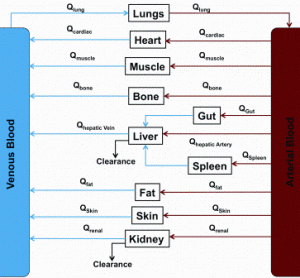
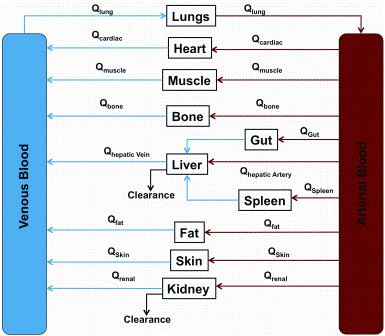
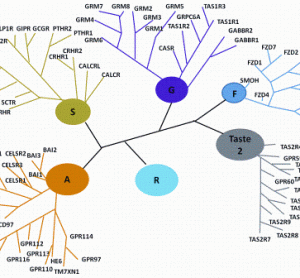
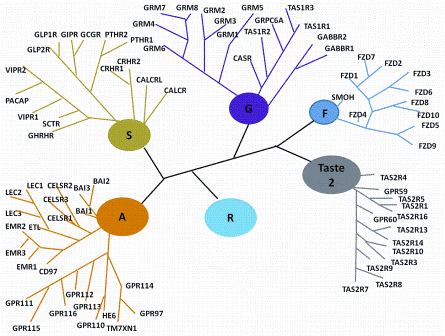
Preparing biocompatible materials for non-permanent medical devices
22 October 2012 | By Vinod B. Damodaran and Jessica M. Joslin, Department of Chemistry, Colorado State University and Melissa M. Reynolds, Department of Chemistry and School of Biomedical Engineering, Colorado State University
Biodegradable polymers comprise an important class of biomaterials due to their ability to satisfy short-term requirements for medical applications where a permanent implant is not required. However, current biodegradable polymers suffer from undesirable chemical properties that lead to improper elimination from the body and potentially toxic by-products. Additionally, medical polymers…