Evaluating drug safety
Posted: 7 March 2005 |
Metabonomics has been defined as the “the quantitative measurement of the time-related multi-parametric metabolic response of living systems to pathophysiological stimuli or genetic modification”1. In simple terms it is a powerful tool for assessing the metabolic component of systemic response.
Metabonomics has been defined as the “the quantitative measurement of the time-related multi-parametric metabolic response of living systems to pathophysiological stimuli or genetic modification”1. In simple terms it is a powerful tool for assessing the metabolic component of systemic response.
Metabonomics has been defined as the “the quantitative measurement of the time-related multi-parametric metabolic response of living systems to pathophysiological stimuli or genetic modification”1. In simple terms it is a powerful tool for assessing the metabolic component of systemic response.
Rather than replace proteomics and genomics, the real power of the technology will lie in synergistic application with its sister technologies. This review will briefly highlight the analytical and biological requirements for successful metabonomics experiments, focusing on the use of the technology for preclinical safety assessment.
Analytical tools for metabonomics
Metabonomics requires the measurement of multiple time-dependent parameters that reflect the biochemical state of an organism. These parameters generally take the form of endogenous small molecule concentrations in a readily available peripheral fluid such as urine or serum (and sometimes tissues or other less readily available fluids). By evaluating how metabolite levels change in comparative experiments (e.g. control vs. drug treated), information about drug efficacy and toxicity can be obtained. The ability to measure as many of these as possible, as rapidly as possible, has spawned new and clever applications of stalwart technologies, such as nuclear magnetic resonance (NMR) and mass spectrometry (MS). Although other analytical approaches have been applied, it is these two that are by far the most widely used in metabonomics applications. In addition to being powerful for identifying specific components in complex mixtures, both NMR and MS can be used to measure patterns of molecular changes without the necessity of assignment of specific molecules that are changing. This can be useful for pattern recognition-base screening approaches3. There are numerous reviews that cover technical aspects and metabonomics applications of both NMR4-6 and MS techniques7-10. Both have their strengths and limitations and their utility in metabonomics will be discussed briefly below. The concept of metabonomics grew from work that focused on the application of (NMR) spectroscopy to study biofluids that began in the early 1980’s11,12. NMR technology proved to be particularly useful because of the rich biomolecular information contained in a simple high-field proton NMR spectrum and the relative ease of sample preparation. This, combined with contemporary advancements in NMR technology during the past two decades (including magnets up to 15 T and cryogenically cooled probes), enabled analysis of increasingly complicated biological systems, including whole biological fluids and tissues. An example of a high field NMR spectrum of rat urine is shown in Figure 1. More than 100 known components can be readily identified by inspection in such a spectrum, based on the proton NMR frequency pattern characteristic of each molecule5.
There are several powerful advantages to NMR spectroscopy that include:
- Signal intensity that is proportional to concentration
- Frequency or chemical shift patterns that are unique to specific molecules
- Linear dynamic range of >108
- Independence of response to matrix
- Dynamical properties of molecules such as ionisation state, diffusion, conformational equilibrium and intermolecular interactions that are measurable through a variety of NMR experiments.
NMR suffers from high cost (with a basic system costing in excess of one million US dollars) and poor sensitivity, generally requiring at least micrograms of analyte for detection under conditions used in most metabonomics applications. In the same time-scale, mass spectrometry (MS) has developed into a tool for analysis of components in complex biological matrices including low molecular weight metabolites and proteins9. Important MS developments in the fields of metabonomics include accurate mass capability and soft ionisation techniques (e.g. electrospray) that allow reduced ambiguity in compound identification and fluidic sample introduction approaches, respectively10. MS is a much more diverse tool than NMR as there are many combinations of sample introduction, ionisation and detection approaches that ultimately yield analytical systems with quite distinct capabilities and which provide different types of information. In general, MS has several intrinsic advantages that include high sensitivity (often thousands of times more sensitive than NMR) and relatively low cost. Downsides to MS include:
- Selective ionisation – an analyte must ionise to be detected and not all molecules ionise under all conditions
- Molecular fragmentation – unstable molecules can disintegrate during ionisation and complicate identifications
- Ion suppression – when more than one species enters the ionisation chamber, there is competition for ionisation energy. This can dramatically impact interpretation of detector responses relative to actual analyte amounts present. Interfering species can be co-eluting molecules, salts or even solvent.
Another major development has been the use of MS as a highly sensitive and selective detector used in conjunction with separation techniques. The most widely used hyphenated MS systems employ either gas chromatography (GC) or high performance liquid chromatography (HPLC) as a front-end separation technique9,13. These two chromatographic interfaces are complimentary and should both be employed when the most thorough metabolic information is desired.
Data analysis
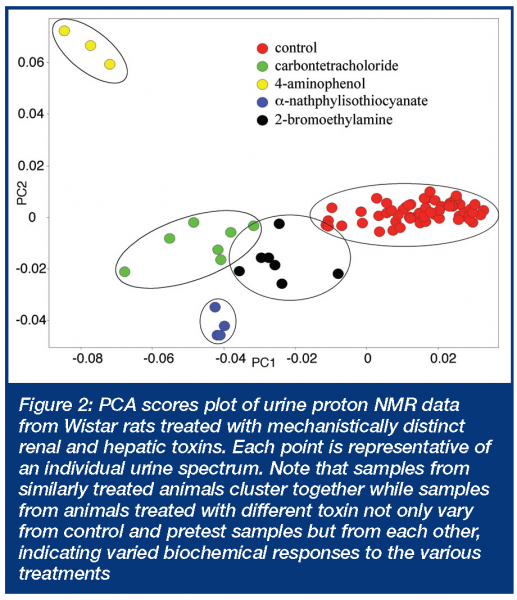
Metabonomics data generated by analytical approaches such as those described above produce massive amounts of data. For example, in a typical study involving 32 rats and 10 time points, an NMR data file can contain upwards of 500 Mbytes of raw and processed data and a chromatography-hyphenated MS data set can be 5 Gbytes or more depending on how many methods are employed10. In both cases, tens or hundreds of thousands of spectral peaks must be measured. This presents a real challenge, since there are currently no commercially available software packages that can seamlessly integrate processing and analysis of such massive and disparate data. To address this challenge, pattern recognition approaches have been applied to both NMR and MS data. Fortunately, once the data is acquired, checked for quality and processed, it is relatively straightforward to express the data in reduced form. For NMR, this would be frequency vs. intensity data and for GC-MS or LC-MS, retention time + mass vs. intensity. Most instrument manufacturers allow such data sets to be exported in a common format such as ACSII. In the case of NMR data, this would provide tens of thousands of data pairs for each spectrum, corresponding to one data pair per point in the digitised NMR spectrum. A commonly used approach for NMR data is to further reduce the dimensionality by integrating over discrete frequency ranges (e.g. 0.04 ppm) within the NMR spectrum, to produce several hundred data points per spectrum. This ‘binning’ approach has the advantage of smoothing the data to account for small frequency shifts in individual component responses and producing a manageable sized data file at the expense of a dramatic reduction in spectral resolution. Such a data file is readily operated on using commercially available software for conducting pattern recognition. Unsupervised approaches, such as principal component analysis (PCA), are commonly used to identify patterns or clusters within such data sets. If the variation between groups is greater than the variation within groups, the data will be segregated in multidimensional or principal component (PC) space (Figure 2). In such cases it is possible to create PC models of normal and abnormal states. The PC coordinates capture the major systematic variation within the data set and the latent variables that define the PC coordinates contain the frequency regions (translatable to biofluid constituents) that are responsible for differentiation of the samples in PC space. PCA is only one of many different pattern recognition approaches that have been applied to develop sophisticated classification models using NMR14-16 and MS13 approaches.
Metabonomics from a toxicology perspective
The power of metabonomics for toxicologic applications lies in the ability to non-invasively assess biochemical response to a compound over the whole time course from pretest to peak effect, through reversal or progression. In practice, serum (or plasma) and urine are the samples of choice with urine providing the potential for continuous sample collection via appropriately designed metabolism cages in preclinical applications. This capability enables reduced animal usage in in vivo studies, as a single animal can be used to collect data from multiple time points. Additionally, temporal variation in response can be identified on an individual animal basis. This further reduces animal requirements as what has frequently been ascribed to ‘interanimal variability’ is, in many cases, unrecognised temporal variation that is missed since continuous sampling is not possible for most typical toxicity endpoints (e.g clinical pathology, histopathology, etc).
Practical application in toxicology studies
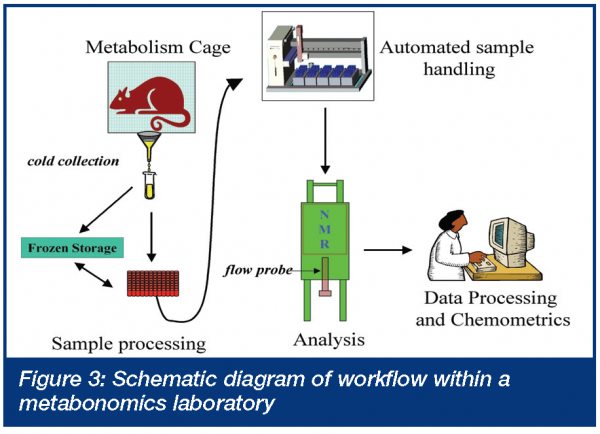
As metabonomics is non-invasive, it can typically be added to existing in vivo pharmacology models or safety studies without the requirement for additional animals or bulk drug. The practical aspects of conducting metabonomics experiments have been reviewed17. Briefly, a typical protocol calls for continuous urine collection (usually in 24 hour time increments) from one day before the initial dose to the termination of the experiment (Generally ≤ 7 days). Longer studies can be run, but urine is usually then collected over defined intervals rather than continuously. Serum (or any other biofluid of interest) can be collected for metabonomic analysis, however, the number of samples per animal will be limited by practical and ethical concerns. Satellite animals can be used for more detailed serum or other biofluid profiles, however, reduced animal requirements and individual animal temporal profiles, are then sacrificed. A typical workflow arrangement of a metabonomic laboratory is presented in Figure 3. Key points include well-controlled sample collection procedures; automated sample processing; automated sample analysis and as much as possible automated data evaluation. It is the last step of the process – data interpretation – that takes the most significant amount of time, in practice. While metabonomics experiments themselves are easy to add to existing studies and models, extreme care must be taken in protocol design and conduct as metabonomics is exquisitely sensitive to protocol details that are frequently overlooked. Metabonomics readily distinguishes metabolic profiles of samples from rats of different strains18 and ages19, at different stages of estrus or from samples collected at different points in the diurnal cycle20. Additionally, vehicles are frequently not as innocuous as we would like to believe21 and either have direct metabolic consequences or simply show up as interferants in the biofluid under study. While this high level of sensitivity may seem disadvantageous, it actually demonstrates the power of the technology to identify systemic issues in animal studies that have always been present but have been ignored, either because we were unaware of them or unable to measure them.
Metabonomics in practice
Compared to proteomics and genomics, metabonomics is relatively new and is only now receiving the evaluation it deserves within the pharmaceutical industry. Within the pharmaceutical industry, metabonomics has had its greatest impact in the area of preclinical safety assessment (toxicology). Within the toxicological setting, three broad application areas can be identified:
- Screening
- Biomarkers
- Mechanisms
One of the earliest attempts to harness the power of the technology was in the arena of rapid throughput screening of candidate therapeutics3,17. Indeed the Consortium on Metabonomics in Toxicology (COMET), a research consortium between six pharmaceutical companies and Imperial College in London, was established for just that purpose22. Figure 2 demonstrates how various renal and hepatic toxins are readily separated in principal component space indicative of differing biochemical responses to these mechanistically distinct toxins. In practice, generating predictive models for generic ‘white powder’ screens (i.e. screening compounds with unknown toxicity) has proven difficult. Although the goal of metabonomics as a white powder screening tool has not been abandoned, it has been tempered by the realisation that systemic responses to toxic effects frequently obscure identification and localisation of biochemical changes to specific target organs. Still, there have been some notable successes. For example, metabonomic evaluation of urine samples has been shown to be quite useful for identifying vascular toxicity, ‘vasculitis’ in rats, a pathology that previously required histopathologic demonstration23. Biomarkers are a trendy topic within various groups within the pharmaceutical industry and toxicology is no exception. Systemic response to toxins complicates the search for biomarkers as it does the search for screens. Therefore, careful (and sceptical) review of the data is required. Despite this requirement, metabonomics has been able to identify some useful biomarkers. Early on, triethylamine and dimethylamine were proposed as region specific biomarkers of nephrotoxicity24. Phenylacetyl glycine was proposed as a biomarker of drug-induced phospholipidosis25 and urinary dicarboxylic aciduria was suggested as a biomarker of impaired fatty acid metabolism26. Mechanisms of toxicity are considered the ultimate achievement in toxicology investigations. A mechanism will always provide a biomarker, although that biomarker may not always prove feasible. Metabonomics has made several notable contributions to mechanistic research. Slim et al demonstrated that phosphodiesterase 4 (PDE4) inhibitor associated vasculitis induced metabolic changes were not the indirect of inflammation but were associated with the vascular lesion itself27. The heretofore unexplained neurotoxic effects of hydrazine were attributed to increased levels of 2-aminoadipate28 and Mortishire-Smith identified disrupted dicarboxylic acid handling as mechanism of impaired fatty acid metabolism which is a common observation with many hepatotoxic pharmaceutical compounds26.
Conclusion
The interest in metabonomics by the pharmaceutical industry is growing exponentially, demonstrated by the increasing rate of publication within this emerging field. In particular, the technology is having a significant impact in the arena of preclinical safety assessment. While human clinical applications are rapidly expanding, the use of the technology in the pharmaceutical clinical trial has not been widespread. This can be anticipated to change as advantages of metabonomics over its sister omic technologies become obvious to pharmaceutical clinicians. Metabonomics is not superior to genomics or proteomics, it simply provides a different level of information. The advantages of the technology include the fact that peripheral samples (urine and blood) can be used and the analytical platform is inherently stable. One only need run a cross-laboratory experiment with a microarray platform to understand why analytical reproducibility is a significant concern. However, the future of the technology will not be based on a competitive analysis of the omic technologies, but on the inherent synergy of a multi-omic approach. These multi-omic (panomic) investigations are just now entering the literature29-32 and we can only anticipate the explosion in basic biochemical understanding that these approaches will provide. 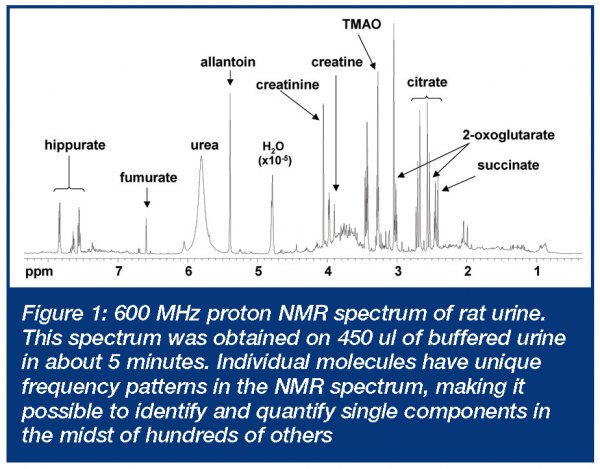



References
- Nicholson, J.K., Lindon, J.C. & Holmes, E. ‘Metabonomics’: understanding the metabolic responses of living systems to pathophysiological stimuli via multivariate statistical analysis of biological NMR spectroscopic data. Xenobiotica 29, 1181-9 (1999).
- Robertson, D.G. Metabonomics in Toxicology: A Review. Toxicological Sciences In Press (2005).
- Robosky, L.C., Robertson, D.G., Baker, J.D., Rane, S. & Reily, M.D. In vivo toxicity screening programs using metabonomics. Comb Chem High Throughput Screen 5, 651-62 (2002).
- Lindon, J.C., Holmes, E., Bollard, M.E., Stanley, E.G. & Nicholson, J.K. Metabonomics technologies and their applications in physiological monitoring, drug safety assessment and disease diagnosis. Biomarkers 9, 1-31 (2004).
- Reily, M.D.and Lindon, J.C. Metabonomics technologies and their applications in physiological monitoring, drug safety assessment and disease diagnosis. in Metabonomics in Safety Assessment (ed. D.G Robertson, J.C.L., E. Holmes and J.K. Nicholson) (Taylor & Francis, New York, 2005), in press.
- Lindon, J.C., Nicholson J.K. and Everett J.R. NMR spectroscopy of biofluids. Ann. Rep. NMR Spect. 38, 1-88 (1999).
- Fernie, A.R., Trethewey, R.N., Krotzky, A.J. & Willmitzer, L. Metabolite profiling: from diagnostics to systems biology. Nat Rev Mol Cell Biol 5, 763-9 (2004).
- Harrigan, G.G., and Goodacre R. (ed.) Metabolic Profiling: Its role in Biomarker Discovery and Gene Function Analysis, 335 (Kluwer Academic Publishers, Boston, 2003).
- Fiehn, O. Combining genomics, metabolome analysis, and biochemical modeling to understand metabolic networks. Comp. Func. Genom. 2, 155-168 (2001).
- van dr Greef, J., van der Heijden, R., and Verheij, E.R. The role of mass spectrometry in systems biology: Data processing and identification strategies in metabolomics. Advances in Mass Spectrometry 16, 145-165 (2004).
- Nicholson, J.K., Buckingham, M.J. & Sadler, P.J. High resolution 1H n.m.r. studies of vertebrate blood and plasma. Biochem J 211, 605-15 (1983).
- Bales, J.R., Higham, D.P., Howe, I., Nicholson, J.K. & Sadler, P.J. Use of high-resolution proton nuclear magnetic resonance spectroscopy for rapid multi-component analysis of urine. Clin Chem 30, 426-32 (1984).
- Plumb, R.S. et al. Metabonomics: the use of electrospray mass spectrometry coupled to reversed-phase liquid chromatography shows potential for the screening of rat urine in drug development. Rapid Commun Mass Spectrom 16, 1991-6 (2002).
- Nicholson, J.K., Connelly, J., Lindon, J.C. & Holmes, E. Metabonomics: a platform for studying drug toxicity and gene function. Nat Rev Drug Discov 1, 153-61 (2002).
- Griffin, J.L. Metabonomics: NMR spectroscopy and pattern recognition analysis of body fluids and tissues for characterisation of xenobiotic toxicity and disease diagnosis. Curr Opin Chem Biol 7, 648-54 (2003).
- Cloarec, O. et al. Evaluation of the orthogonal projection on latent structure model limitations caused by chemical shift variability and improved visualization of biomarker changes in (1)h NMR spectroscopic metabonomic studies. Anal Chem 77, 517-26 (2005).
- Robertson, D.G., Reily, M.D., Lindon, J.C., Holmes, E., and Nicholson, J.K. Metabonomic Technology as a Tool for Rapid Throughput In Vivo Toxicity Screening. in Comprehensive Toxicology, Vol. 14 (ed. Vanden Heuvel, J.P., Perdew, G.J., Mattes W.B., and Greenlee W.F.) 583-610 (Elsevier Science BV, Amsterdam, 2002).
- Holmes, E., Nicholson, J.K. & Tranter, G. Metabonomic characterization of genetic variations in toxicological and metabolic responses using probabilistic neural networks. Chem Res Toxicol 14, 182-91 (2001).
- Robertson, D.G. et al. Metabonomics: evaluation of nuclear magnetic resonance (NMR) and pattern recognition technology for rapid in vivo screening of liver and kidney toxicants. Toxicol Sci 57, 326-37 (2000).
- Bollard, M.E. et al. Investigations into biochemical changes due to diurnal variation and estrus cycle in female rats using high-resolution (1)H NMR spectroscopy of urine and pattern recognition. Anal Biochem 295, 194-202 (2001).
- Beckwith-Hall, B.M. et al. NMR-based metabonomic studies on the biochemical effects of commonly used drug carrier vehicles in the rat. Chem Res Toxicol 15, 1136-41 (2002).
- Lindon, J.C. et al. Contemporary issues in toxicology the role of metabonomics in toxicology and its evaluation by the COMET project. Toxicol Appl Pharmacol 187, 137-46 (2003).
- Robertson, D.G., Reily, M.D., Albassam, M. & Dethloff, L.A. Metabonomic assessment of vasculitis in rats. Cardiovasc Toxicol 1, 7-19 (2001).
- Anthony, M.L., Gartland, K.P., Beddell, C.R., Lindon, J.C. & Nicholson, J.K. Studies of the biochemical toxicology of uranyl nitrate in the rat. Arch Toxicol 68, 43-53 (1994).
- Nicholls, A.W., Nicholson, J.K., Haselden, J.K., and Waterfield C.J. A metabonomic approach to the investigation of drug-induced phospholipidosis: An NMR spectroscopy and pattern recognition study. Biomarkers 5, 410-423 (2000).
- Mortishire-Smith, R.J. et al. Use of metabonomics to identify impaired fatty acid metabolism as the mechanism of a drug-induced toxicity. Chem Res Toxicol 17, 165-73 (2004).
- Slim, R.M. et al. Effect of dexamethasone on the metabonomics profile associated with phosphodiesterase inhibitor-induced vascular lesions in rats. Toxicol Appl Pharmacol 183, 108-9 (2002).
- Nicholls, A.W. et al. Metabonomic investigations into hydrazine toxicity in the rat. Chem Res Toxicol 14, 975-87 (2001).
- Coen, M. et al. Integrated application of transcriptomics and metabonomics yields new insight into the toxicity due to paracetamol in the mouse. J Pharm Biomed Anal 35, 93-105 (2004).
- Kleno, T.G., Kiehr, B., Baunsgaard, D., and Sidelmann, U.G. Combination of ‘omics’ data to investigate the mechanism(s) of hydrazine-induced hepatotoxicity in rats and to identify potential biomarkers. Biomarkers 9, 116-138 (2004).
- Verhoeckx, K.C. et al. A combination of proteomics, principal component analysis and transcriptomics is a powerful tool for the identification of biomarkers for macrophage maturation in the U937 cell line. Proteomics 4, 1014-28 (2004).
- Verhoeckx, K.C. et al. Characterization of anti-inflammatory compounds using transcriptomics, proteomics, and metabolomics in combination with multivariate data analysis. Int Immunopharmacol 4, 1499-514 (2004).




