MicroRNAs for high-throughput functional analysis
Posted: 19 June 2008 | Vytaute Starkuviene, Urte Sudžiuviene· and Holger Erfle, VIROQUANT/BIOQUANT, University of Heidelberg, Germany | No comments yet
MicroRNAs (miRNAs) are a class of small non-coding RNA molecules, which are potent post-transcriptional gene expression regulators. They have been shown to participate in the regulation of numerous cellular processes, the list of which is still growing. miRNAs affect numerous targets that can be determined by direct experiments or predicted by bioinformatics approaches, and are presented in several online databases. Feasibility of miRNA for high-throughput experimentation is becoming possible due to the availability of commercially produced molecules, which are able to alter the levels of endogenous miRNAs. miRNA functional analysis will help to validate predicted targets and reveal the role of these small molecules in biological pathways. miRNAs have a high potential to be used as a new gene expression regulating reagent for microscopy based assays.
MicroRNAs (miRNAs) are a class of small non-coding RNA molecules, which are potent post-transcriptional gene expression regulators. They have been shown to participate in the regulation of numerous cellular processes, the list of which is still growing. miRNAs affect numerous targets that can be determined by direct experiments or predicted by bioinformatics approaches, and are presented in several online databases. Feasibility of miRNA for high-throughput experimentation is becoming possible due to the availability of commercially produced molecules, which are able to alter the levels of endogenous miRNAs. miRNA functional analysis will help to validate predicted targets and reveal the role of these small molecules in biological pathways. miRNAs have a high potential to be used as a new gene expression regulating reagent for microscopy based assays.
MicroRNAs (miRNAs) are a class of small non-coding RNA molecules, which are potent post-transcriptional gene expression regulators. They have been shown to participate in the regulation of numerous cellular processes, the list of which is still growing. miRNAs affect numerous targets that can be determined by direct experiments or predicted by bioinformatics approaches, and are presented in several online databases. Feasibility of miRNA for high-throughput experimentation is becoming possible due to the availability of commercially produced molecules, which are able to alter the levels of endogenous miRNAs. miRNA functional analysis will help to validate predicted targets and reveal the role of these small molecules in biological pathways. miRNAs have a high potential to be used as a new gene expression regulating reagent for microscopy based assays.
The discovery of the RNA interference (RNAi) phenomenon a decade ago1 not only truly revolutionised our understanding about gene regulation processes, but also became a superior tool for unravelling the function of unknown genes2,3. Around the same time as RNAi, miRNA(s) were discovered4, and were shown to be potent regulators of gene expression in viruses, animals and plants5,6. Their regulatory effect is exerted through the pairing to complementing mRNA molecules and inducing translational repression7. Most miRNAs are thought to make imperfect matching8, what is largely the reason for their ability to regulate expression of numerous mRNAs simultaneously9.
It is well documented that expression levels of miRNA molecules are changing under diverse cellular conditions10, in case of neoplastic transformations11 and during viral infections12. Therefore, the functional analysis of doses-dependant behaviour of miRNAs would not only be of interest in understanding cellular circuits involved, but also would be indispensable in identifying novel pharmaceutical targets and setting new diagnostic procedures.
Prediction of miRNA targets and miRNA databases
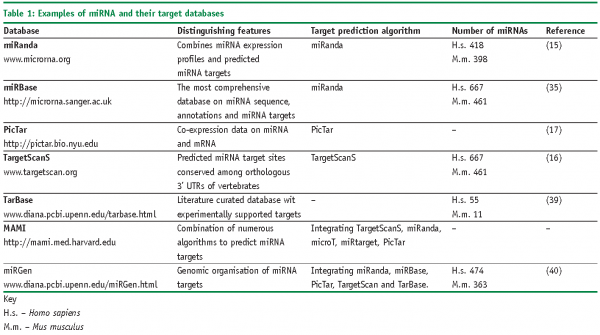
Imperfect matching of miRNAs to their targets not only induces complex biological responses, but also makes their target prediction to be a daunting task13, which essentially relies on a quality of genome sequence annotation, particularly on that of 3’UTR14. Several different strategies are used to identify the targets of miRNAs specifically. Some of the algorithms provide target predictions largely based on sequence complementarity between mRNA and the seed region of miRNA (miRanda15, TargetScan16 or PicTar17). Other target predictions methods prefer calculations of energetically favourable hybridisation between microRNA and target mRNA (RNAhybrid18, StarMir19). However all of them predict hundreds if not thousands of targets, which potentially can be affected by a given miRNA. More specifying information can be obtained from resources, which record experimentally validated miRNA targets, flanking miRNA sequences, species conservation of miRNA-mRNA binding sites, co-expression of miRNA and their potential targets and many other features (Table 1). Still, inference the biological processes regulated by these molecules is difficult without the well-controlled experimental system.
Currently, several miRNA databases are publicly available (Table 1). Some of them utilise original algorithms (miRanda, PicTar), some of them unify the information from diverse publicly available sources (miRGen, MAMI). Sometimes, only few model organisms are taken into consideration, for instance targets only for human, mouse and rat miRNA are present in miRanda database. On contrary, information on targets from almost 24 species is presented in miRBase – the most comprehensive depository of miRNA data to date. Unfortunately, as the approaches are different, numbers of miRNA targets and targets themselves differ considerably across the databases. This situation is still one of the most hindering factors in applying miRNA for high-throughput functional analysis and remains to be improved in the future.
Finally, the numbers of miRNA are varying across databases. That again depends upon the strategy of how miRNA was predicted, isolated and/or validated. Initially, miRNA was defined as being expressed as a distinct transcript, which can be detected by experimental means (e.g., Northern blot) and originating from the characteristic hairpin precursor, which is processed by Dicer20. Ideally, miRNA sequence and structure should be conserved evolutionary. In general, these characteristics are applied by identifying miRNAs by experimental approaches (size-fractionation of cDNA libraries, cloning and sequencing)21 and by bioinformatics approaches (e.g., secondary sequence prediction, thermodynamics of hairpins, genomic location and clustering)22. The discrepancy in miRNA numbers originates from the numerous factors, such as abundance of miRNA molecule and sequence modifications, difficulty to associate hairpin structures specifically to miRNA than to the rest of genome, just to mention a few.
Currently, the number of miRNAs is several times higher as anticipated originally23, and is increasing at a constant rate. However, the actual numbers do not hit more than 600 molecules in human genome, for instance. That is still several times less than theoretically predicted numbers ranging from several to ten of thousands miRNAs24, making miRNA a promising reagent for truly high-throughput studies.
miRNAs as a tool for functional analysis
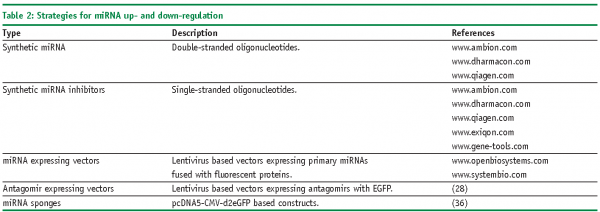
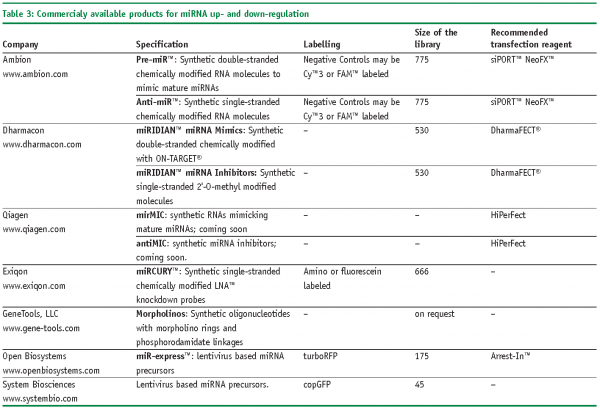
Though the number of predicted miRNAs is expanding, only few of them are functionally annotated25. However, the situation might be changing fairly soon as commercially produced libraries of reagents are available for manipulation of endogenous levels of miRNAs (Table 2 and Table 3). In general, two types of reagents are available: encoded by recombinant vectors and chemically synthesised. Chemically synthesised molecules are offered by a number of well established producers as individual molecules, specified sub-sets or genome-wide libraries. As a rule they mimic the composition and, consequently, the behaviour of mature miRNA. Synthetic oligonucleotides are degraded rapidly in cells what restricts their usage for transient transfection experiments. A somewhat lower variety of products covers vectors encoding miRNAs; in difference from chemically synthesised ones they are preferential for stable-transfection experiments, transfection of primary and non-dividing cells, and largely encode primary miRNAs. As an example of such producers Open Biosystems can be mentioned, which offer a set of more than 170 lentivirus based vectors encoding primary miRNAs with their natural flanking sequences. That enables to maintain endogenous regulation of miRNA biogenesis. Besides commercial products, some academia researches constructed libraries of miRNA-related reagents. For instance, retroviral constructs or UAS-DsRed plasmid based constructs encoding several hundreds of miRNA precursors were published recently26,27. Specific miRNA loss-of function phenotypes can be investigated with lentivirus-mediated antagomir expression cassettes28.
Functional reagents based on miRNA are already as superior as siRNAs due to their stability9, versatility of their practical applications, and due the ease these reagents can be transfected.
Gain-of-function and loss-of-function of miRNA
‘Gain-of-function’ miRNA phenotypes can be raised by transfecting cells with chemically synthesised double-stranded oligonucleotides or plasmids encoding sequences of miRNA. In case of the latter constructs, fluorescent proteins are frequently attached to monitor expression efficiency and to separate transfected cells from untransfected cells (e.g., miR-expressTM).
Specific down-regulation of miRNA can be achieved by chemically synthesised oligonucleotides, so called antagomirs, which bind to the mature miRNAs. As a rule, these are single-stranded RNA molecules carrying diverse modifications. One of the first described were 2-O-Methyl oligoribonucleotides (2-O-Me-RNA), which were used to identify components of the miRNA biogenesis pathway29. 2-O-Me-RNA was described to bind RNA probes faster and with higher temperature melting curves30. Such type of oligonucleotides is now offered by Dharmacon under the commercial name miRIDIANTM miRNA inhibitors.Another type of modification, so called LNATM (Locked Nucleic Acids) are based on a methylene bridge connecting the 2’-O atom with the 4’-C atom in the ribose, which increases the pairing of the oligonucleotide to the complementary target31. As such they provide resistance against cellular nucleases and are, therefore, considered to be more effective in terms of the length of down-regulation experiments. Selective LNATM chemistry-based oligonucleotides are offered by several companies, for instance Santaris Pharma (www.santaris.com) to target liver specific miRNA-122 for curing Hepatitis C and Hypercholesterolaemia. Somewhat less applicable for high-throughput studies are morpholinos – chemically modified RNA molecules with a 6-members morpholino ring instead of a 5-members ribose32. They bind complementary RNA molecules and prevent their further actions by steric blockage33. Due to their unnatural backbone, morpholinos are totally resistant to nuclease cleavage34 and can be used for prolonged down-regulation experiments, e.g., in developmental biology. However, a serious drawback of these molecules is their relatively inefficient delivery into cells if to compare with regular RNAi molecules.
Instead of using ‘one after one’ inhibition strategy, the down-regulation of functional families of miRNA might be more efficient in unravelling miRNA targets, particularly when 30-40% of miRNA are expressed as clusters35. To achieve this goal, so called ‘sponges’ constructs were created on a basis of pcDNA5-eGFP vectors (www.invitrogen.com). These molecules, encoding divergent target sequences of miRNAs, were shown to ‘soak’ a pool of respective endogenous miRNAs as efficiently as chemical oligonucleotides36.
miRNA functional screens
The potential of miRNAs to alter diverse biological processes and the availability of their manipulation tools opens the needs and possibilities to perform comprehensive functional screens in order to position them into one or another functional context. Frequently, the functional behaviour of miRNAs is derived from the studies of expression profiling followed by appropriate functional experiments37. The regulatory role of miRNA could be successfully deciphered by direct functional assessment as reported by26, where a library of over-expressed miRNAs was used to substitute the loss of p53 for neoplastic transformations. In the study of Ovcharenko and colleagues 200738, 187 synthetic miRNAs representing the majority of the human miRNAs that were known to exist at the time were tested to participate in apoptosis pathway. 34 of these miRNAs led to a differential caspase-3 activation phenotype. Quantitative cell-based screens were implied to test the changes in transcription regulation, namely Wingless pathway27. Cell proliferation was recently assessed by modulating levels of selected set of miRNA molecules28.
Although, above-mentioned examples are still sparse in the field of miRNA research, cell-based functional screens has a high potential to reveal biologically relevant miRNA activities and targets.
Outlook
The availability of reagents to modulate the endogenous levels of miRNAs, steadily improving algorithms for prediction of their targets, developments of high-throughput functional analysis techniques together with quantitative data analysis and modelling approaches is already now opening new avenues for understanding regulatory cellular networks on a yet unprecedented level.



References
- A. Fire et el: Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature, 391(6669), pp.806-11, 1998.
- L. Cullen, G. Arndt: Genome-wide screening for gene function using RNAi in mammalian cells. Immunol Cell Biol, 83(3): pp.217-23, 2005.
- C. Echeverri, N. Perrimon: High-throughput RNAi screening in cultured cells: a user’s guide. Nat Rev Genet, 7(5): pp.373-84, 2006.
- M. Lagos-Quintana et al: Identification of novel genes coding for small expressed RNAs. Science, 294(5543): pp.853-8, 2001.
- J. Carrington, V. Ambros: Role of microRNAs in plant and animal development. Science, 18;301(5631): pp. 336-8, 2003.
- S. Pfeffer et al: Identification of virus-encoded microRNAs. Science,30;304(5671): pp.734-6, 2004.
- D. Bartel: MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 23;116(2): pp. 281-97, 2004.
- J. Brennecke et al: Principles of microRNA-target recognition. PLoS Biol. 3(3):e85, 2005.
- L. Lim et al: Microarray analysis shows that some microRNAs down regulate large numbers of target mRNAs. Nature. 17;433(7027): pp.769-73, 2005.
- J. Neilson et al: Dynamic regulation of miRNA expression in ordered stages of cellular development. Genes Dev. 1;21(5): pp.578-89, 2007.
- G. Calin, C. Croce: MicroRNA signatures in human cancers. Nat Rev Cancer. 6(11): pp.857-66, 2006.
- R. Triboulet et al. Suppression of microRNA-silencing pathway by HIV-1 during virus replication. Science. 16;315(5818): pp.1579-82, 2007.
- N. Smalheiser, V. Torvik: Complications in mammalian microRNA target prediction. Methods Mol Biol. 342: pp.115-27, 2006.
- P Sethupathy et al: A guide through present computational approaches for the identification of mammalian microRNA targets. Nat Methods. 3(11): pp.881-6, 2006.
- B. John et al: Human MicroRNA targets. PLoS Biol. 2(11):e363, 2004.
- B. Lewis et al: Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell. 14;120(1): pp.15-20, 2005.
- A Krek et al:Combinatorial microRNA target predictions. Nat Genet. 37(5): pp.495-500, 2005.
- M Rehmsmeier et al: Fast and effective prediction of microRNA/target duplexes. RNA. 10(10): pp.1507-17, 2004.
- Y. Ding et al: RNA secondary structure prediction by centroids in a Boltzmann weighted ensemble. RNA. 11(8): pp.1157-66, 2005.
- K. Saito et al: Processing of pre-microRNAs by the Dicer-1-Loquacious complex in Drosophila cells. PLoS Biol. 3(7):e235, 2005.
- A. Aravin, T. Tuschl: Identification and characterization of small RNAs involved in RNA silencing. FEBS Lett. 31;579(26): pp.5830-40, 2005.
- I. Bentwich: Prediction and validation of microRNAs and their targets. FEBS Lett. 31;579(26): pp.5904-10, 2005.
- L. Lim et al: Vertebrate microRNA genes. Science. 7;299(5612): p.1540, 2003.
- K. Miranda et al: A pattern-based method for the identification of MicroRNA binding sites and their corresponding heteroduplexes. Cell. 22;126(6): pp.1203-17, 2006.
- N. Bushati, S. Cohen: microRNA functions. Annu Rev Cell Dev Biol.23:175-205, 2007.
- P. Voorhoeve et al: A genetic screen implicates miRNA-372 and miRNA-373 as oncogenes in testicular germ cell tumors. Cell. 24;124(6): pp.1169-81, 2006.
- S. Silver et al: Functional screening identifies miR-315 as a potent activator of Wingless signaling. Proc Natl Acad Sci U S A. 2007 Nov 13;104(46):18151-6.
- M. Scherr et al: Lentivirus-mediated antagomir expression for specific inhibition of miRNA function. Nucleic Acids Res. 35(22):e149, 2007.
- G. Hutvágner et al: Sequence-specific inhibition of small RNA function. PLoS Biol. 2(4):E98, 2004.
- Majlessi M et al: Advantages of 2′-O-methyl oligoribonucleotide probes for detecting RNA targets. Nucleic Acids Res. 1;26(9): pp.2224-9, 2008.
- M. Petersen et al: The conformations of locked nucleic acids (LNA). J Mol Recognit. 13(1): pp.44-53, 2000.
- J. Summerton, D. Weller: Morpholino antisense oligomers: design, preparation, and properties. Antisense Nucleic Acid Drug Dev. 7(3): pp.187-95, 1997.
- Kang H et al: Stacking interactions of ApA analogues with modified backbones. Biopolymers. 32(10): pp.1351-63, 1992.
- R. Hudziak et al: Resistance of morpholino phosphorodiamidate oligomers to enzymatic degradation. Antisense Nucleic Acid Drug Dev. 6(4): pp.267-72, 1996.
- S. Griffiths-Jones et al: miRBase: tools for microRNA genomics. Nucleic Acids Res. 36(Database issue):D154-8, 2008.
- M.Ebert et al: MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells. Nat Methods. 4(9): pp.721-6, 2007.
- F. Meng et al: Involvement of human micro-RNA in growth and response to chemotherapy in human cholangiocarcinoma cell lines. Gastroenterology. 130(7): pp.2113-29, 2006.
- D. Ovcharenko et al: Genome-scale microRNA and small interfering RNA screens identify small RNA modulators of TRAIL-induced apoptosis pathway. Cancer Res. 15;67(22): pp.10782-8, 2007.
- P. Sethupathy et al: TarBase: A comprehensive database of experimentally supported animal microRNA targets. RNA. 12(2): pp.192-7, 2006.
- M. Megraw et al: miRGen: a database for the study of animal microRNA genomic organization and function. Nucleic Acids Res. 35(Database issue):D149-55, 2007.




