A perspective from Eli Lilly and Co.
Posted: 23 May 2006 | | No comments yet
Advances in optical imaging methods, personal computer power and cell/molecular biology methodology have merged to form the field of ‘Cellomics’1 also referred to as High Content Cellular Imaging (HCCI). HCCI is a powerful and flexible cell-based assay platform that has the potential to shorten cycle times by broadly impacting the Drug Discovery process from Target Validation/Lead Generation through in vitro support of Clinical Candidates. This article provides an overview of HCCI, contrasts it with conventional cell based assay modalities, and provides general examples of the technology’s effects on the Drug Discovery process at Eli Lilly and Company.
Advances in optical imaging methods, personal computer power and cell/molecular biology methodology have merged to form the field of ‘Cellomics’1 also referred to as High Content Cellular Imaging (HCCI). HCCI is a powerful and flexible cell-based assay platform that has the potential to shorten cycle times by broadly impacting the Drug Discovery process from Target Validation/Lead Generation through in vitro support of Clinical Candidates. This article provides an overview of HCCI, contrasts it with conventional cell based assay modalities, and provides general examples of the technology’s effects on the Drug Discovery process at Eli Lilly and Company.
Advances in optical imaging methods, personal computer power and cell/molecular biology methodology have merged to form the field of ‘Cellomics’1 also referred to as High Content Cellular Imaging (HCCI). HCCI is a powerful and flexible cell-based assay platform that has the potential to shorten cycle times by broadly impacting the Drug Discovery process from Target Validation/Lead Generation through in vitro support of Clinical Candidates. This article provides an overview of HCCI, contrasts it with conventional cell based assay modalities, and provides general examples of the technology’s effects on the Drug Discovery process at Eli Lilly and Company.
What is High Content Cellular Imaging?
In its simplest terms, High Content Cellular Imaging is fluorescence spectroscopy at a sub-cellular spatial resolution. Like conventional spectroscopy, HCCI can quantitatively measure fluorescence intensity, colour, excitation/emission spectra, anisotropy and excited state lifetimes. Significantly, HCCI differs from conventional assay modalities by measuring spectral properties at the level of single cells rather than a single averaged measurement of a collection of cells. Like flow cytometry, HCCI detects spectral heterogeneity of individual cells in a sample and can thereby differentiate sub-populations. High Content Cellular Imaging, unlike fluorescence based cytometry, is not restricted to measuring intensity and colour; the spatial and time resolution of HCCI enables analysis of cell morphology, object size and sub-cellular localisation as a function of time. HCCI is therefore a powerful analytical method that provides quantitative, multiplexed measures of cellular and sub-cellular features that can be utilised to define cellular sub-populations within the sample.
These analytical capabilities of High Content Cellular Imaging are particularly significant in conjunction with advances in cell and molecular biology. These advances include the greater availability of ‘physiologically’ relevant cellular systems and an arsenal of fluorescent labeling methods to detect specific proteins, post-translational protein modification, cellular components (eg DNA, mitochondria, plasma membrane, cytoskeleton, microtubules), and changes in cell function (eg. toxicity, plasma membrane permeability, membrane potential, intracellular Ca, reactive oxygen species) (http://probes. invitrogen.com/handbook/). This fusion between new analytical and cell biology technologies provides an unprecedented ability to manipulate and measure a vast array of signal transduction and cell biology events in medium and high throughput. In addition, Cellular Imaging’s ability to identify and analyse cell sub-populations when combined with advances in stem and primary cell biology can extend these analytical capabilities to rare and more physiologically relevant cell types. The convergence and synergism of these diverse technologies positions HCCI as a powerful and broadly applicable assay platform for cell biology, analogous perhaps to liquid scintillation counting in the mid-20th century. High Content Cellular Imaging promises to be a fertile ground for rapid advances in Drug Discovery, Cell Biology, System Biology, and Chemogenomics.
Cellular Imaging within Eli Lilly and Company
Lilly has recently experienced significant growth in HCCI technology. In 2005 the number of automated HCCI instruments increased from two to eight with the net addition of three instruments planned for 2006. Similarly, the number of ‘power user’ groups has increased and currently includes scientists from various therapeutic areas and core groups from biology and chemistry.
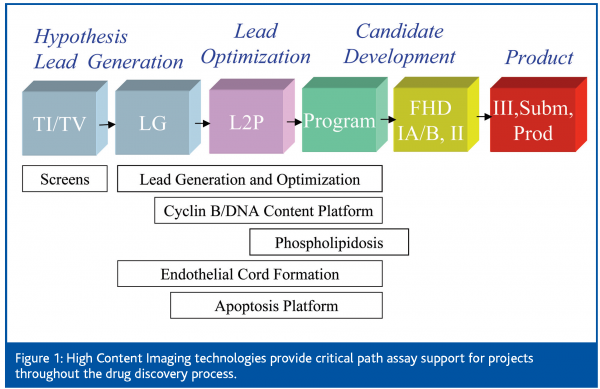
Figure 1 summarises the impact of High Content Cellular Imaging to Eli Lilly’s drug discovery process. In general, the drug discovery process can be conceptualized in four parts. Generation of a therapeutic hypothesis encompasses target identification and validation, providing a linkage of the molecular target or signal pathway to a therapeutic indication. Once a validated target or signal pathway is identified, initial ‘hits’ are identified by MTS or HTS during Lead Generation. Subsequently, potency, efficacy and early ADME characteristics of the most promising hits are optimised by iterative cycles of medicinal chemistry and compound testing in the Lead Optimisation stage. Animal toxicology studies occur early in Candidate Development followed by Product support. At Eli Lilly and Company, HCCI provides critical path, decision making assay support for many projects ranging from Lead Generation to early in vitro Toxicology (Figure 1).
Roles of High Content Cellular Imaging in drug discovery
The pharmaceutical industry’s collective experience in MTS, HTS and quantitative biology has provided the basic tools needed to use HCCI for analysis/visualisation macroscopic cell parameters. At Lilly, in vitro assays used to make critical path decisions require statistical validation by methods and criteria that have been recently published on the NIH Chemical Genomics Web site (http://www.ncgc.nih.gov/guidance/ index.html). Our statistical validation process examines inter-plate and inter-day reproducibility of signal window, Z’ scores, signal uniformity and compound efficacy2,3. Passing criteria are Z’ scores > 0.4 with no indication of systematic signal variation and MSR values of < 3, indicating that an efficacy ratio between two compounds of greater than three fold is statistically significant2.
Lead generation
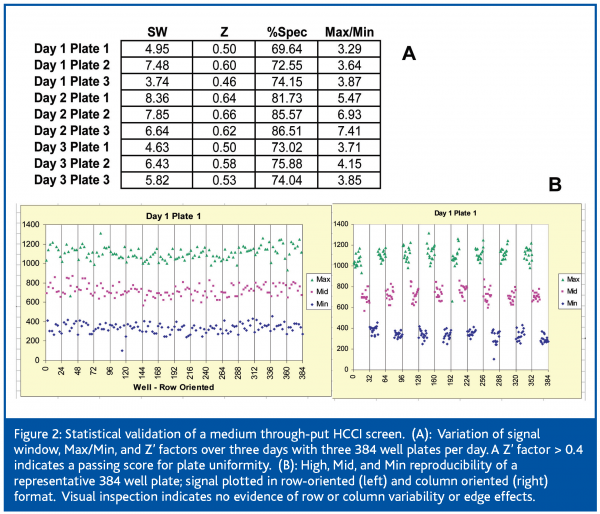
High Content Cellular Imaging has been used to enable both target directed and, more recently, phenotypic or signal pathway screens. HCCI is compatible with a variety of assay modalities utilising either transiently or stably transfected cells lines, 96 or 384 plate formats, and multiplexing up to 4 cell signal parameters. Figure 2 shows representative plate uniformity and assay validation data from an MTS configured by transient transfection of two cDNAs with a single channel output. The summary of plate Z’ scores over the three day validation indicates sufficiently reproducible transfection, cell plating, and signal output with no indication of systematic plate variations (Figure 2).
Lead optimisation
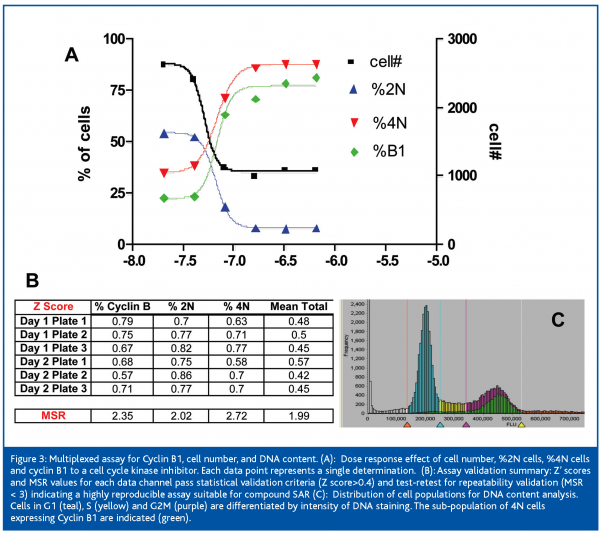
Lilly’s experience has indicated that HCCI is a powerful platform for supporting compound SAR efforts. Figure 3 shows the validation statistics and typical results from an Acumen Explorer assay which simultaneously measures Cyclin B, cell number, and DNA content. Gating of the DNA channel based on fluorescence intensity allows identification of cell populations with the ‘normal’ 2N DNA levels associated with the G1 phase, 4N DNA levels associated with the cell population that has undergone DNA replication but not mitosis (G2 to M), and the cell population undergoing DNA replication (S phase). Cyclin B1 levels can be correlated to specific cell cycle phases at the single cell level while cytotoxicity of compound can be roughly estimated by cell number, nuclear morphology and density changes. Two day validation of %Cyclin B1, %2N, and %4N data resulted in Z’ scores from 0.57 to 0.86 whereas average total cell number passed with Z’ scores ranging from 0.42 to 0.57. Significant to SAR support, the MSR values range from 1.99 to 2.72 indicating that the assay can reliably discriminate compound potency ratios greater than 2 and 2.7 fold, respectively.2
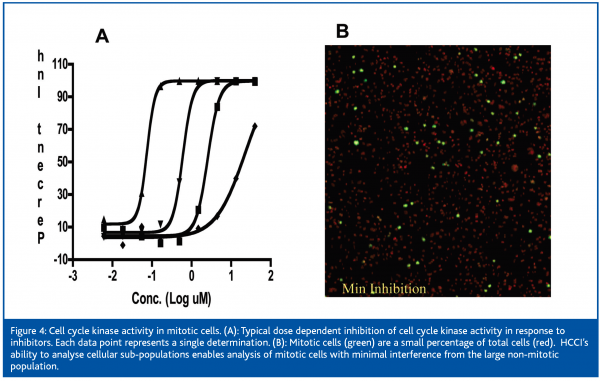
The Acumen Explorer has also been useful in enabling several kinase assays involved in cell cycle regulation. Attempts to develop and validate macroscopic cell based kinase assays using ELISA or cell ELISA based methods were repeatedly unsuccessful (Cox, unpublished). In contrast, HCCI based phosphorylation assays passed plate uniformity and MSR criteria and typically run with one data point per compound concentration (Figure 4). The enhanced HCCI assay performance observed with cycle applications may be due to the small percentage of mitotic cells within the sample. Methods that monitor a signal from the entire well may have low, but finite background in non-mitotic cells (Red, Figure 4). Since this population of cells comprise >90% of the total cells, the overall background signal may obscure the specific signal from the mitotic cells (Green, Figure 4). In contrast, HCCI based assays can gate exclusively on the mitotic cells and monitor changes specifically within this sub-population. In addition to sub-population gating, the Acumen Explorer can rapidly conduct full well scans and thereby increase the number of interrogated cells by at least an order of magnitude as compared to microscope based systems. These instrument features enhance assay reproducibility, minimises plate edge effects, and increases through-put.
High Content Cell Biology
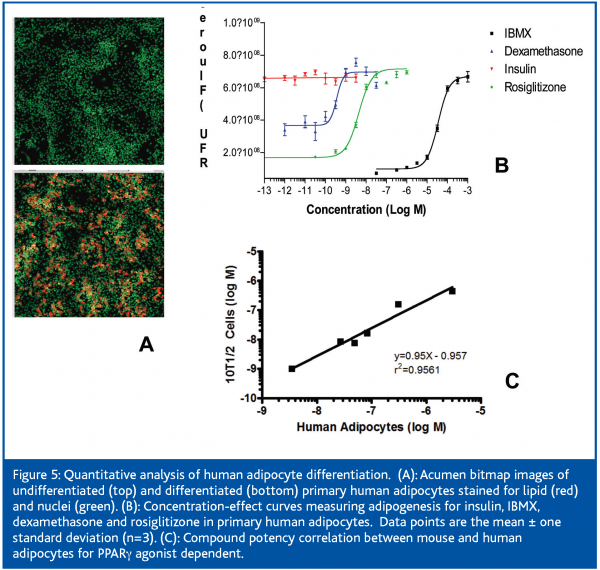
High Content Cellular Imaging assays are particularly useful in later phase drug discovery activities such as mechanism-of-action studies and phenotypic profiling to rank order compounds prior to in vivo testing. For example, diabetes and obesity research projects rely on measuring lipid accumulation in rodent derived adipocytes although species differences in efficacy and potency is a potential issue. Initially, we found that methods for differentiating human adipocytes were not quantitative and amenable to well averaged fluorescence readers. We therefore developed an HCCI assay to quantitate lipid accumulation within the cell mixture resulting from human pre-adipocyte differentiation (Figure 5). Experimental design methods were used to optimise differentiation and media conditions to develop a quantitative adipogenesis assay (Z’ of 0.57; MSR of 1.9). This assay was used to directly compare the potencies of various PPARγ agonists to differentiate rodent and human preadipocytes; the excellent in vitro potency correlation for the PPARγ agonists tested (Figure 5) increases the likelihood of an in vivo correlation.
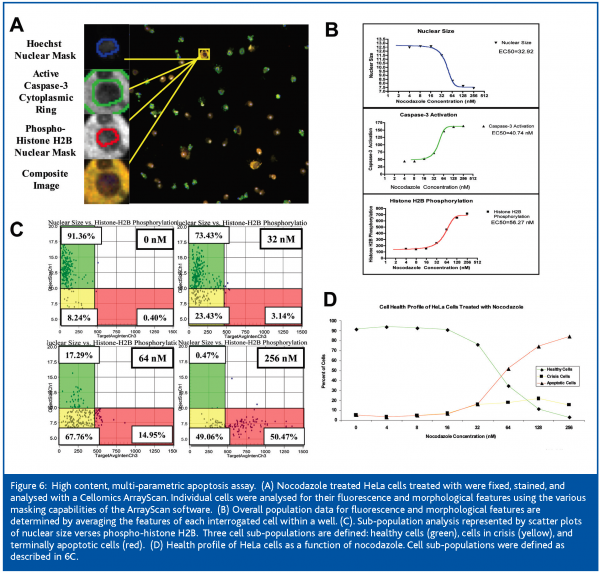
Cellular apoptosis is an important profiling assay for multiple drug discovery efforts. It is a complex biological process which is described by multiple cellular characteristics which need to be correlated with modulation of target activity. We currently employ an apoptosis assay that incorporates three fluorescent markers analyzed on a Cellomics Array Scan VTI. Hoechst 33342 for detection of cells, definition of the nuclear compartment, quantitation of nuclear condensation/fragmentation, and analysis of DNA content; anti-active caspase-3 antibody is used to detect early to intermediate apoptotic events; and anti-phospho-histone H2B assesses terminal apoptosis Analysis of these apoptotic events with software to identify sub-cellular regions, cellular sub-populations, and derive multi-parametric criteria for the categorisation of cells provide information on mechanism of action (Figure 6).
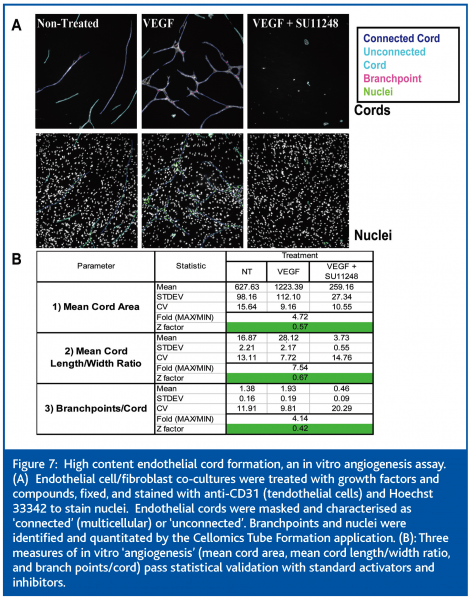
HCCI is also extremely useful for the quantitation of phenotypes involving complex multicellular structures. The formation of pseudo-vessel cords by endothelial cells is an assay used to assess effects on angiogenesis in vitro. We have developed a high content, 96-well cord formation assay that utilises endothelial cells co-cultured with fibroblasts. An anti-CD31 antibody differentially labels the endothelial cells, while Hoechst 33342 is used to stain nuclei. Several morphological features of cord formation are utilized to detect subtle pro- or anti-angiogenic effects of compounds in a medium through-put mode (Figure 7).
In vitro Toxicology
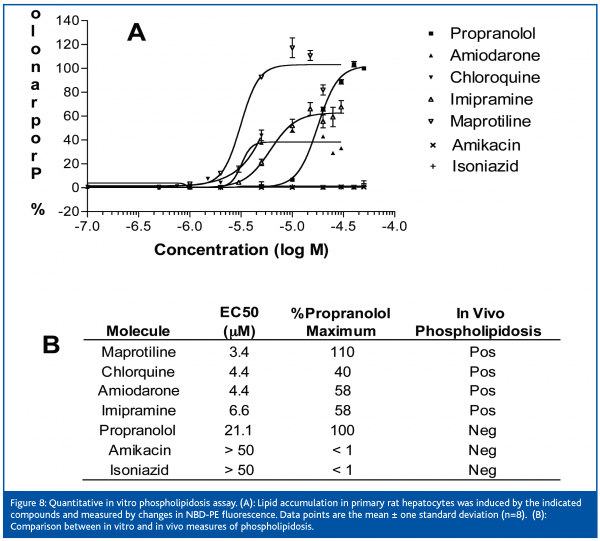
Cationic amphiphilic drugs are known to induce phospholipidosis (PL), the intracellular accumulation of lysosomal phospholipids. PL can differentially affect liver, skeletal muscle, kidney and other tissues by unknown mechanisms and therefore raises potential long-term drug safety concerns. Traditional methods to access PL are qualitative and involve animal studies or manual microscopic inspection. HCCI was used to enable a 96 well, quantitative PL assay (Z’= 0.76) in primary rat hepatocytes. This in vitro assay correlates with in vivo measures of PL using standard compounds (Figure 8) and supports compound scaffold selection and toxicity profiling and for advanced stage projects for the past three years.
Increasing the accuracy of surrogate toxicity tests remains a challenge with a very high potential impact on the drug discovery process. Dr. Peter O’Brien from the Discovery Toxicology department of Pfizer has recently presented exciting progress using HCCI for predictive toxicology4. O’Brien’s group tested over 600 compounds with known human ADME and toxicology data on HepG2 cells while monitoring five parameters of cell health. Compounds were grouped by the number, type and potency of the cell health parameters affected and compared to in vivo data. Based on the in vitro parameters affected, the authors claim to be able to predict high, medium, and low in vivo human toxicology with an 87% correlation4.
Future developments
High Content Cellular Imaging is a powerful cell based assay platform that greatly impacts the drug discovery process by enabling unique assays in rare or complex mixtures of cells. Recently, Eli Lilly and Company has experienced a rapid growth in this technology and has applied HCCI successfully to address critical path problems through out the drug discovery process (Figure 1).
What are the developing user needs as High Content Cellular Imaging expands and becomes more broadly integrated into the drug discovery process? The number and diversity of commercially available HCCI instruments have steadily increased without a concomitant development of appropriate IT systems to effectively store and manage the large amount of image and meta data generated by HCCI (estimated at 7 T bytes/year at Lilly). The cost of servers, IT infrastructure and personnel is a very significant “hidden cost” of HCCI and is frequently limiting. Therefore established HCCI users with operational IT and server solutions may choose to purchase new instruments on the basis of IT compatibility rather than instrument performance. Vendors that lead in IT infrastructure solutions and imaging applications will greatly benefit by making their IT systems compatible with more instrument platforms. Similarly, instrument venders with under developed IT solutions will benefit by using common image and meta data formats to increase the compatibility of their instruments with existing IT solutions.
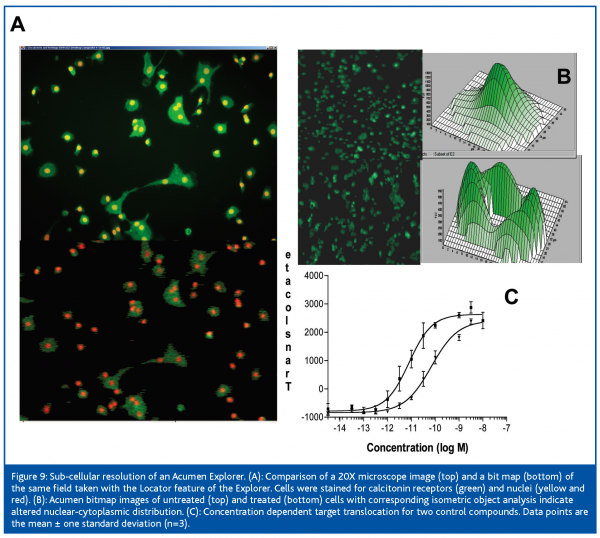
Cost-effective instrument platforms which combine the advantages of microscope based systems and laser scanning systems will improve data collection rates and enhance the statistical quality of assays. We have noted that high resolution bit maps from an Acumen Explorer are similar in appearance to a 20X fluorescence microscope image (Figure 9). Usually regarded as a low resolution technology, current laser scanning devices can quantitate changes in intracellular location of GFP fusion proteins in the experimental systems investigated. Although bitmaps generated by laser scanning devices may not be considered ‘true’ images, they can be analysed and quantitated by conventional image analysis software. The coupled use of high resolution scanning instruments with image analysis software will present significant IT and data storage challenges, but may provide a highly sensitive and cost effective imaging solution which combines sub-cellular spatial resolution with rapid whole well data collection.
The impact of new cell biology technologies are greatly enhanced by HCCI. Recently there has been a steady increase in the availability of stem, progenitor and primary cells. These cellular systems when used with experimental design methods to optimize cell culture conditions and growth surfaces may potentially lead to more physiologically relevant in vitro cell systems. These cellular systems, when used with RNAi approaches, provide investigators an unprecedented ability to manipulate cell biology at genome wide scales. The ensemble of these cell biology technologies is complemented by HCCI’s unique analytical capabilities, such as the multiplexed quantitation of intracellular localisation, cellular sub-populations, and multicellular morphologies. Collectively, these advances in cell biology and high content cellular imaging provide an unprecedented ability to manipulate and measure a vast variety of cellular models.
While the application of High Content Cellular Imaging provides novel avenues of research and contributes to critical path decisions along the drug discovery process (Figure 1), the technology is not currently being used to its full potential. Many HCCI imaging programs measure multiple parameters at the level of individual cells but this data is typically used to enhance the signal to noise ratio of well averaged parameters with less consideration of cellular population distributions or non-parametric analysis methods. HCCI is inherently multi-parametric but the majority of applications utilise data channels independently with infrequent use of multivariate analysis to describe cellular responses. Future development of robust and innovative IT, statistics, and data visualisation tools are needed before HCCI realises its full potential.
Success in the highly competitive, post-genomic environment of the pharmaceutical industry requires innovation and close collaboration between end users, cell biologists, chemists, informatics specialists, statisticians, database designers, and hardware providers. The organisations that effectively incorporate and leverage the full potential of High Content Cellular Imaging will emerge as leaders during the next decade.









Acknowledgements
We thank our colleagues Richard Beckman, Laura Bloem, Lorri Burris, Fred Chadwell, Simon Chen, Jack Eastridge, Mike Esterman, Rachelle Galvin, Jeff Hansen, Phil Iversen, Weiming Li, Diego Medina, David K. Monteith, Ryan Morgan, Julie Pomeroy, Jianyong Shou, Sean Sissons, Jeff Smallwood, Xiaoling Xia, and Xiang Ye for patient collaboration and our various management teams for their support.
References
- Taylor, L. and Wang, Y.L., Fluorescent Labeled Molecules as Probes of the Structure and Function of Living Cells, Nature 284, 405-10, 1980.
- Eastwood, B.K., Farmen, M.K., Iverson, P.W., Craft, T.J., Smallwood, J.K., Garbison, K.E., DeLapp, N.W., and Smith, G.F. The Minimum Significant Ratio: A Statistical Parameter to Characterize the Reproducibility of Potency Estimates from Concentration-Response Assays and Estimation of Replicate-Experiment Studies, J. Biomol. Screen 2006, in press.
- Iverson, P.W., Eastwood, B.J., Sittampalam, G.S., and Cox, K.L., Comparison of Assay Performance Measures in Screening Assays: Signal Window, Z’ Factor, and Assay Variability Ratio Biomol. Screen 2006, in press.
- O’Brien, P., Developing in vitro Cytotoxicity Assays for HCS, presentation at High Content Screening: Information Rich Cellular Analysis to Accelerate your Drug Discovery, October 19, 2005, London, UK




