Featured content
Gaelic Labs: Pharma is increasingly focused on supply chain security
General Manager Brian Morrissey discuss his company’s acquisition of fellow Beta-Lactam manufacturer Athlone Laboratories and its implications for an evolving industry.
Harnessing AI for more efficient clinical trials
During this virtual panel, industry experts discussed practical examples and considerations for applying AI in pharma’s clinical trials.
Practical considerations for aseptic gowning in contamination control strategies
Watch this webinar as we examine the main personnel-related factors that could lead to particle/microbial contamination risks of the product and outline practical measures that can mitigate these risks.
Complying with USP Chapters 41 and 1251 revisions for pharma quality control
Join this webinar to gain insight about the changes to United States Pharmacopeia (USP) General Chapters 41 and 1251 on balance requirements for quality control.
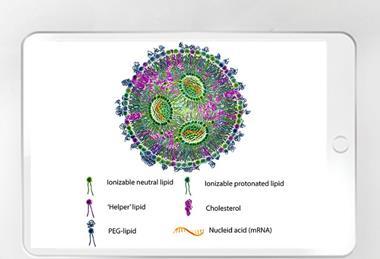
Navigating evolving clinical and regulatory expectations as mRNA outgrows its vaccine roots
Jianbo Diao, PhD, Director of Bioanalytical Services, WuXi AppTec, discusses the importance for mRNA therapy developers of adopting precise and flexible analytical strategies for future pipeline success.
Opinion and interviews
AI in pharmaceutical development: hype or panacea?
Dave Elder reflects on the array of applications for AI to facilitate successful drug development, though proof remains elusive.
- Previous
- Next
Unlock exclusive industry insights
- Bookmark articles and resources to access anytime
- Enjoy free access to industry leading resources, webinars and insights
- Stay informed with the latest news and breakthroughs in drug discovery
- Receive updates and recommendations tailored to your research interests
Webinars
- Previous
- Next
Whitepapers
- Previous
- Next