Featured content
Real-time Raman spectroscopy closes bioprocess control measurement gap
Manual sampling cycles leave bioreactor control decisions hours behind culture dynamics. Process Raman spectroscopy offers continuous, real-time measurement of critical parameters, enabling true closed-loop control in perfusion and continuous bioprocessing applications where measurement lag directly impacts yield and product quality.
European Pharmaceutical Review Issue 1 2026
EPR Issue 1 includes articles on manufacturing, drug delivery, environmental monitoring, analytical techniques and more.
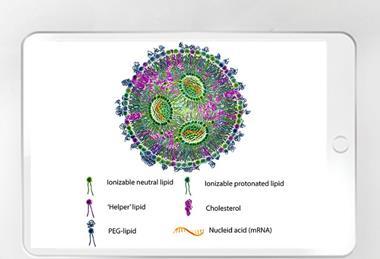
Optimising the downstream process in lentiviral vector manufacturing
Sartorius BIA Separations illustrates a promising alternative approach for efficient lentiviral vector purification that delivers a scalable, GMP-ready biomanufacturing solution.
Optimising efficiency and yield through bioprocessing automation
Join this webinar to explore how process Raman spectroscopy supports bioprocessing automation across upstream and downstream workflows, helping teams increase yields, improve process efficiency, and ensure more consistent product quality.
Opinion and interviews
Real-time Raman spectroscopy closes bioprocess control measurement gap
Manual sampling cycles leave bioreactor control decisions hours behind culture dynamics. Process Raman spectroscopy offers continuous, real-time measurement of critical parameters, enabling true closed-loop control in perfusion and continuous bioprocessing applications where measurement lag directly impacts yield and product quality.
- Previous
- Next
Unlock exclusive industry insights
- Bookmark articles and resources to access anytime
- Enjoy free access to industry leading resources, webinars and insights
- Stay informed with the latest news and breakthroughs in drug discovery
- Receive updates and recommendations tailored to your research interests
Webinars
- Previous
- Next
Whitepapers
- Previous
- Next